1.1 – Alcohol Metabolism
Available @ notion too. Click here to view!
An overview of ethanol metabolism
Ethanol is a polar liquid.
It is water soluble – this means it can pass through biological membranes (like cell membranes) just as easily as water molecules can.
Ethanol is practically insoluble in fats and oils.
The equilibrium concentration of alcohol depends on:
- the relative water content of an area
- the rate of blood flow
- the tissue mass.
Alcohol will distribute from the blood to all the tissues in proportion to the relative content of water.
Difference between male and female ethanol consumption
Women have an overall smaller volume of fluid in their body compared to males. This means the overall alcohol concentration in the body will be higher in females than males (less water content →higher alcohol concentration).
Absorption of alcohol from intestine to the bloodstream
Alcohol is absorbed in the digestive system – this includes the stomach and the intestines. This is through passive diffusion, where alcohol molecules move from an area of high concentration (intestine walls after consumption) to an area of low concentration (the bloodstream).
Peak alcohol levels is greater if it is all consumed in one large dose than many smaller doses. This may be because there are more molecules to metabolise at once.
Peak alcohol levels are also higher when having alcohol in an empty stomach. The presence of food reduces gastric emptying, as it slows down the consumption of alcohol.
Alcohol Metabolism
90% of alcohol is metabolised in the liver. The remainder is excreted through urination or passed out through the breath.
The enzymes involved in oxidation of alcohol to acetaldehyde include ADH (Alcohol dehydrogenase) and, to a lesser extent, cytochrome P450 dependent oxidising system in the liver, and catalase in the brain.
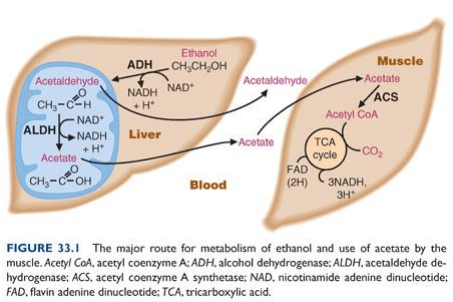
Note: acetaldehyde is toxic, whereas acetate is not. This means ALDH is a significant enzyme for converting the metabolite to a safer form.
Acetate is converted to Acetyl CoA in muscles to enter the Krebb’s cycle, which is then used for energy
Significance of Cytochrome P450
Assists in converting alcohol to acetaldehyde
More than 100 different cytochrome P450 enzymes exist within these 10 genome families.
The one with the highest affinity to alcohol is CYP2E1.
Note: CYP2E1 can metabolise alcohol and therefore reduce alcohol concentration in the body however it can also have negative consequences. This is because as alcohol concentration decreases, the amount of toxic acetaldehyde increases, which increases the risk of hepatic injury.
Cytochrome enzymes have a higher affinity for alcohol than ADH enzymes.
Alcohol Metabolism – Liver Damage
Alcohol induced liver damage is a side effect of alcohol usage.
This can be in the form of:
- Fatty liver
- Alcohol induced hepatitis
- Liver cirrhosis (Fibrosis of liver tissue)
ALDH – usually in charge of preventing the toxic product from accumulating. Even so, when acetaldehyde accumulates in great amounts this can be sufficient enough to cause liver damage.
Mechanisms of liver damage:
Fatty Liver: prevention of fatty acid oxidation and accumulation of triacylglycerol synthesis
Alcohol induced hepatitis: generation of free radicals from acetaldehyde causes inflammation of hepatocytes
Liver cirrhosis: permanent damage to hepatocytes causes the scarring and accumulation of fibrous tissue. This is followed by abnormal blood flow and loss of liver function.
Drug for heavy drinkers – Disulfram
Supports alcohol use disorder by producing a sensitivity response to ethanol.
It works by blocking the oxidation of alcohol at the acetaldehyde stage
The accumulation of acetaldehyde causes many symptoms such as nausea, vomiting, palpitations, blurred vision and dyspnea.
