Vaccines were always a controversy. The discovery of the vaccine was highly criticised by society, and even 300 years later there is still a fiery debate with different members of society on opposite sides, leading to significant impact in the department of public health. It is actually a fact that 23% of Americans eligible for vaccinations had chosen not to take it.
The COVID-19 vaccine is somewhat described as the major turning point in our journey through this ongoing pandemic. But variants after variants come and attack regions of the earth, which all have different properties, making lockdown the only final remaining solution. How well has this vaccine worked to improve the pandemic?
It is important to first of all acknowledge how much work has been put in to even produce these vaccines. Pfizer had put into place a Phase 3 clinical trial to determine safety and effectiveness starting from July 2020. The vaccine was released and given to the first patient in the UK 5 months later. It’s important to evaluate the statistics, provided by Pfizer themselves, of the trial conducted for this vaccination.
The Phase 3 clinical trial was designed to determine if the Pfizer-BioNTech COVID-19 vaccine is safe and effective in preventing COVID-19 disease.
This trial began July 27, 2020, and completed enrollment of 46,331 participants in January 2021.
On November 18, Pfizer and BioNTech announced that, after conducting the primary efficacy analysis, their mRNA-based COVID-19 vaccine met all of the study’s primary efficacy endpoints.
On December 2, 2020, the Medicines & Healthcare Products Regulatory Agency (MHRA) in the U.K. authorized the Pfizer-BioNTech COVID-19 vaccine for emergency use, marking the first Emergency Use Authorization following a worldwide Phase 3 trial of a vaccine to help fight the pandemic.
Shortly after on December 11, 2020, the U.S. Food and Drug Administration (FDA) authorized the Pfizer-BioNTech COVID-19 vaccine for emergency use.
An Overview of the Pfizer Phase 3 trial:
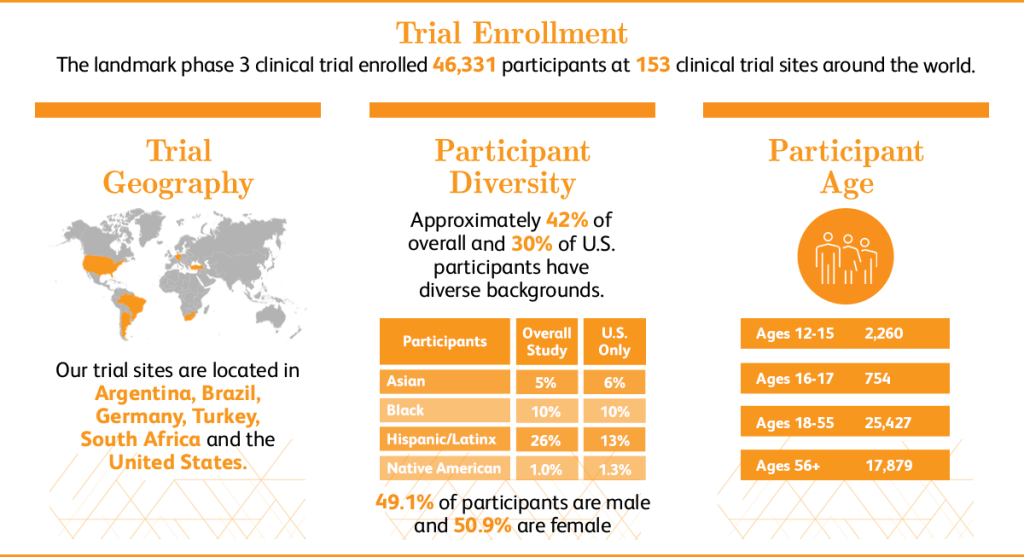
As clearly evident from the statistics:
- The number of participants is a high proportion
- There is an even distribution between males and females that participated in the trial
- There is a wide range of ages varying from 12-56+, with a higher proportion of adults that participted
- Participants are from regions around the world
Overall this provides assurance and reliability of the steps they had taken to evaluate safety and effectiveness, as there is no evidence it has been only targeted to a specific group of people.
It has been evident that 1 in 4 people who took the Pfizer vaccine has experienced mild symptoms, with headache, fatigue and tenderness being the most commonly reported. These side effects have been claimed to last only around 1-2 days. Some adverse effects have also been reported, including a severe allergic reaction (anaphylactic shock) and red rashes too. These adverse effects are however extremely rare. In most cases of adverse effects, it is recommended a person doesn’t further get the vaccine booster anymore. If the adverse effects weren’t that severe, however, it is possible for them to get another dose (depending on what side effects were experienced).
Let’s now take a close look at the AstraZeneca vaccine. This is the vaccine which people think is the most dangerous due to the death of patients from blood-clotting, as a side effect of its intake. In the UK, AstraZeneca is now used less particularly for the elderly as there were a rare few cases of patients that had died from blood-clotting as an effect.
The AstraZeneca Trial was a Phase 3 trial which had actually started from April 2020. In August the trial was put to a hold for a short while due to the reporting of one patient with adverse neurological effects, however not much information was released about this case. These are the details AstraZeneca has provided about the Phase 3 trial they conducted:
- 32,449 participants accruing 141 symptomatic cases of COVID-19.
- The trial had a 2:1 randomisation of vaccine to placebo.
- 79% were white/Caucasian, 8% black/African American, 4% native American and 4% Asian, and 22% of participants were Hispanic.
- Approximately 20% of participants were 65 years and over, and approximately 60% had comorbidities associated with an increased risk for progression of severe COVID-19, such as diabetes, severe obesity or cardiac disease.
Although the number of participants who took AstraZeneca is less than that of Pfizer, its still quite a high number. There was a high number of people that were white/caucasian in comparison to all the other ethnicities, although there is evident variety. Not much information was given on the different ages that took part, although a good fifth was over 65s. This makes sense as this category was more likely to get affected severely by the virus.
Timeline for the development of the AstraZeneca Vaccine
April 30, 2020
AstraZeneca’s vaccine uses a modified chimpanzee virus to deliver instructions to the body to make the coronavirus spike protein. This in turn triggers an immune response.
May 18, 2020
AstraZeneca signs a deal with the U.K. government to supply up to 30 million doses of the vaccine by as soon as September and to provide 100 million doses by the end of the year.
September 1, 2020
The company begins its large-scale Phase III human clinical trial of the vaccine in the U.S.
November 9, 2020
Pfizer beats AstraZeneca to become the first major pharmaceutical company to announce the results of its Phase III clinical trials. The company says its jab is more than 90% effective, increasing the pressure on AstraZeneca to show that its vaccine is also highly effective.
November 19, 2020
Oxford researchers publish more data in The Lancet showing that the vaccine they are developing with AstraZeneca elicits a strong immune response in older adults. The finding is important because the elderly are most at risk from COVID-19.
November 23, 2020
AstraZeneca and the University of Oxford announce the initial results of the Phase III clinical trials in the U.K., Brazil, and South Africa. The study finds the vaccine safe and effective, but the results are presented confusingly, combining data from trials conducted with different protocols and giving out three different efficacy figures. The company says that a “blended average” showed the vaccine was about 70% effective.
December 30, 2020
The U.K. becomes the first country to approve AstraZeneca’s vaccine.
January 29, 2021
The European Medicines Authority approves the AstraZeneca vaccine for use in all adults, but adds that the initial clinical trials did not include enough people over the age of 55 to be certain of the effectiveness in that age group.
March 11, 2021
Denmark stops rollout of the AstraZeneca vaccine because of concerns about dangerous blood clotting among some who received the jab. The company’s stock falls on the news. AstraZeneca insists that the number of clotting incidents reported so far are actually fewer than would occur normally for the millions of people who have now been given the inoculation worldwide so far.
March 16, 2021
A number of other European countries, including Germany, France, and Italy, also suspend distribution of the AstraZeneca vaccine amid concerns about clotting even as the European Medicines Agency and the World Health Organization say the vaccine is safe.
March 22, 2021
AstraZeneca announces interim results from its U.S. clinical trial, saying the vaccine was 79% effective against symptomatic COVID-19 and 100% effective against hospitalization and death. The company hopes the results of the 30,000-person study in the U.S. and South America will finally clear the way for the vaccine to be approved by the U.S. FDA.
Side effects of the vaccine are generally the exact same as those of the Pfizer vaccine. However, there has been an increased controversy about the AstraZeneca vaccine after a small proportion of people died from thrombosis. It has not yet been clear from the NHS that the AstraZeneca vaccine is linked to this blood clotting, however AstraZeneca claims the following:
“The vaccine was well tolerated, and the independent data safety monitoring board (DSMB) identified no safety concerns related to the vaccine. The DSMB conducted a specific review of thrombotic events, as well as cerebral venous sinus thrombosis (CVST) with the assistance of an independent neurologist. The DSMB found no increased risk of thrombosis or events characterised by thrombosis among the 21,583 participants receiving at least one dose of the vaccine. The specific search for CVST found no events in this trial.”
In the UK, AstraZeneca is not being given to patients too old or too young, or those who have a higher possibility of blood clotting. It is important to remember that the blood clotting events happened to a very small proportion of the people who got vaccinated with AstraZeneca, should the vaccine be the cause of it. And like all other vaccines, a small proportion of the population will receive mild side effects – this is why your healthcare provider will always ask for allergies and any other health conditions to determine whether you are likely to get adverse side effects.
Myths and facts about the COVID-19 Vaccinations
- Myth 1: The ingredients of the pfizer vaccine are dangerous.
This is not true. Although it is a chemical solution, it consists of ingredients that would be found in everyday foods – including fats, sugars and salts. Seeing how the process of dilution is done pre-injecting, a simple solution of NaCl (basic salt) is added to the concentrated original mixture- a lot less complex than many may have thought!
- Myth 2: Natural Immunity from being attacked by the virus is better and stronger than that of the vaccine
This isn’t true. In fact, getting the vaccine is a lot safer as you will experience overall less side effects than you would do by being infected! It still produces the exact same antibody response, and there is evidence suggesting that it causes added protection to those who have already been affected by COVID-19. So why go through the trouble of getting infected first!
- Myth 3: COVID-19 vaccines cause variants
COVID-19 vaccines actually prevent new variants from emerging. This is because it prevents the amount of people that can get affected from it, killing it and hence giving it a lower chance of having the opportunity to variate.
- Myth 4: The vaccine contains microchips
The COVID-19 vaccine does not contain microchips. Vaccines are only for your protection and safety, not to monitor or track movement. Was this the case, there would have been an easier way to implement microchips than vaccinating everyone.
- Myth 5: The vaccine can alter DNA
This vaccine doesn’t anyhow alter your DNA or mRNA. It merely triggers the development of cells to produce an immune response against the virus, without incorporating itself into the DNA. The vaccine’s other ingredients are discarded as waste as a normal form of body function.
- Myth 6: The COVID-19 vaccine isn’t even considered as a vaccination.
This vaccine doesn’t inject viruses into your body like some other vaccinations do. However, it does inject mRNA which triggers immune cells to recognise the ‘spike protein’ (protein that identifies the cell). This recognition means if your body does come in contact with the virus, they will already have memory lymphocytes to fight against the virus.
Vaccines have shown effectiveness in this pandemic. Since the first vaccination programme was put into place in the UK. With 87.7% of the population receiving at least 1 dose of the vaccine, the pandemic was almost something people didn’t worry about during the summer months of 2021. It was even reported that most people that were getting hospitalised with COVID were those that were not jabbed, which had also encouraged many to take the vaccine afterwards. But yet again another variant has evolved which seems to be affecting those even with 2 doses. Getting a third dose will provide further immunity to the variant so it is highly encouraged for everyone to get one more dose. After all, there seems to be no other way out of this pandemic, as seen so far. Take the vaccine and help protect the world from this pandemic. Even if it’s your first dose it’s not too late!

Leave a comment